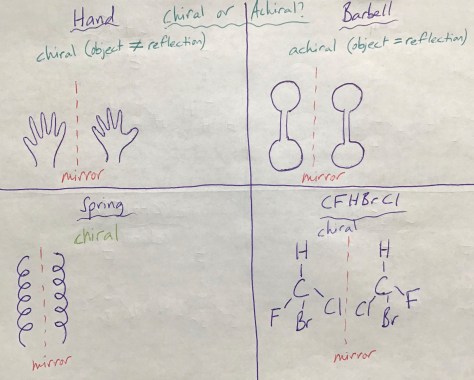
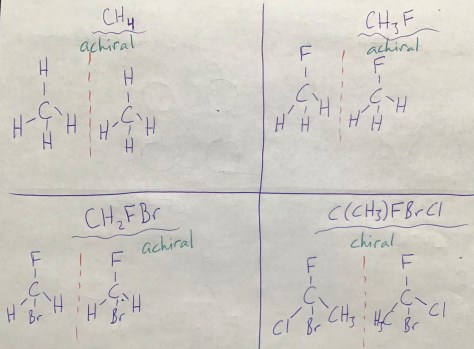
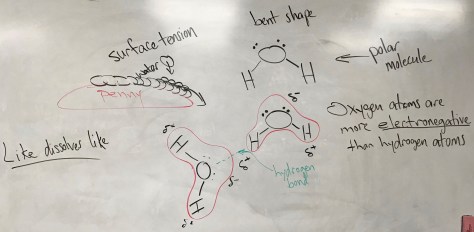
The final lesson of Unit 2 explores how amino acids connect to make proteins. The Lesson 48 PowerPoint includes the vocabulary terms of amino acid and protein. Lesson 48 connects with Lesson 47, as amino acids are chiral molecules. Notably, all of the 20 different amino acids in human proteins are “left-handed” (as opposed to the mirror-image “right-handed” isomers), meaning they all have the L conformation (L for laevus, Latin for “left”) rather than the D conformation (D for dexter, Latin for “right”). Students will work in pairs to complete the Lesson 48 Worksheet, learning about the properties of amino acids and how they bind together to form proteins. For more on the D and L convention, click on the picture below.
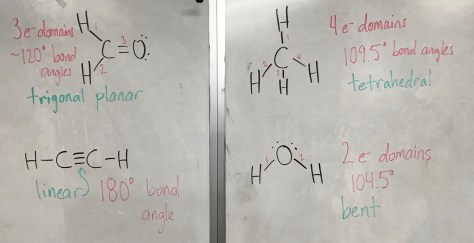
As noted previously, there are 20 different amino acids. All amino acids share the same base structure of a central carbon atom bound to a carboxylic acid (-COOH), an amino group (-NH2), and a hydrogen (H). The central carbon is also bound to an R group, with R indicating any one of the 20 different amino acid structures. The structures each have different physical properties. When individual amino acids link together, a polypeptide chain is formed (and a molecule of water is removed as each new amino acid is linked to the chain). The polypeptide chain, composed of a string of amino acids, folds into a particular shape determined by the interactions of all of the amino acids. The shape of a protein determines its function in the body. Mr. Anderson of Bozeman Science has a fantastic video explaining the nature of proteins:
For students looking for a good review of Central Dogma (DNA > RNA > Protein > Trait), the Crash Course Biology video DNA, Hot Pockets, & The Longest Word Ever is a good resource:
Finally, for students with access to a home computer, the Fold.It website will have you folding proteins in no time!
Homework:
- Read Lesson 48 in the textbook. Login via hs.saplinglearning.com and enter your username and password.
- Write notes for Lesson 48 and work through the practice problems at the end of Lesson 48
- Please ask questions about anything from Lesson 48 you do not yet fully understand.









You must be logged in to post a comment.