With 10th grade students on the Career Field Trip, we took the class period to review what we have learned so far in Chapter 13. Students had the opportunity to ask questions, practice balancing equations via a worksheet and models, of use computers to work through the Balancing Chemical Equations and/or the Chemical Changes simulations on the ExploreLearning Gizmos website.
Category Archives: Chemistry
Balancing Chemical Equations
Class began with the ChemCatalyst from the Lesson 72 PowerPoint. With the lesson focusing on balancing chemical equations, we worked through the ChemCatalyst equation with a focus on differentiating coefficients and subscripts (slides 11 and 12). We practiced balancing a few more equations and then students received the Lesson 72 Worksheet to work through. For homework, students were assigned questions 2 and 4 from the textbook.
For students who would like additional instruction around balancing equations and enjoy learning by watching videos, I recommend Tyler DeWitt’s videos Introduction to Balancing Equations (above) and Balancing Chemical Equation Practice Problems (below):
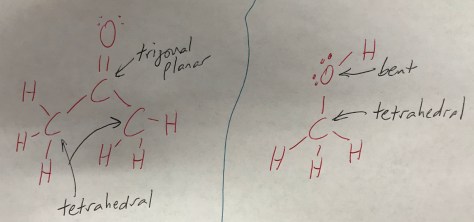
Physical vs Chemical Change
For Lesson 70, we began by recalling the lab yesterday and then observing the outcome of the following reaction: Cobalt (II) chloride (a solid with a rich blue color) + water (clear liquid) reacts to form a pink liquid. An image of the reaction is in the Lesson 70 PowerPoint. We discussed the difference between physical and chemical changes, and then used the remainder of the short class period to complete the Lesson 70 Worksheet. For homework, students were assigned textbook questions 4 and 5 from Lesson 70.
For students who were absent or would like additional review, please watch the video below from Mr. Anderson of Bozeman Science:
Chemical Equations
To begin Unit 4, we worked through the first few slides of the Lesson 68 PowerPoint and students then watched a video showing the chemical reaction written on the worksheet. Students then received the Lesson 68 Worksheet and Toxin Card Deck, along with a graphic organizer for analyzing the card deck and then they worked in groups of four to complete the activity.
Review Comparing Mitosis and Meiosis
With our next quiz rapidly approaching (tomorrow!) we used the first half of class to compare mitosis and meiosis. Our work is shown below:
After the review, students had the opportunity to complete the dihybrid cross Gizmo. Those who finished received a copy of a dihybrid cross practice worksheet for further review. A copy of the completed front side of the worksheet can be viewed by clicking here.
Ask all the questions!
Our review of Units 1 and 2 culminated today. Notes from both classes are provided below:
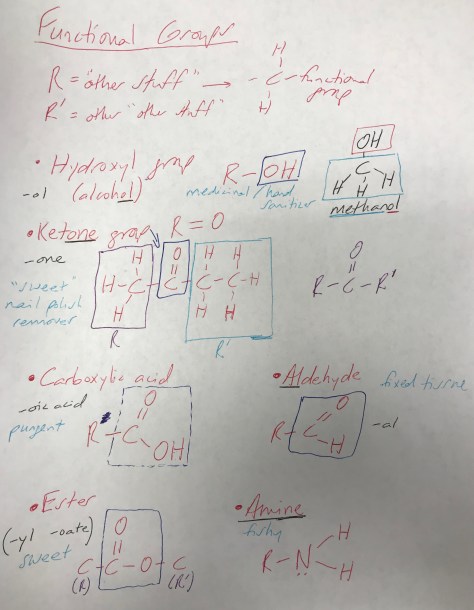
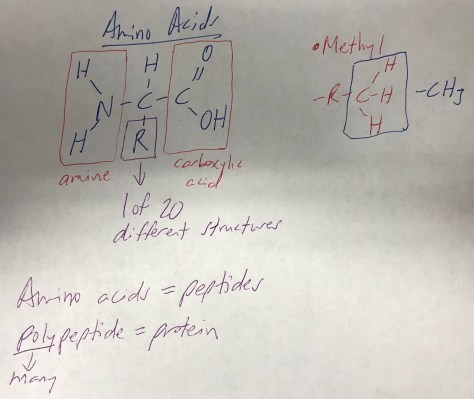
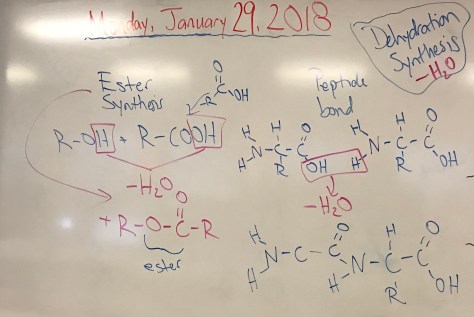

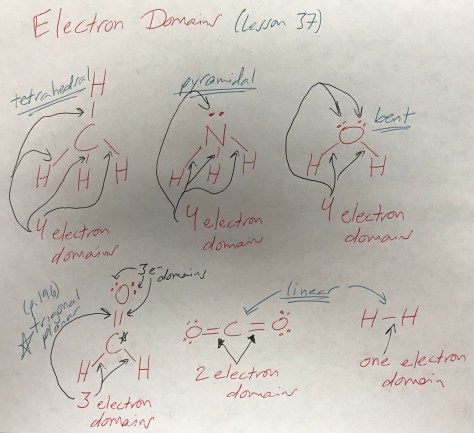
Mirror-Image Isomers
We began with the Lesson 47 PowerPoint ChemCatalyst to help get students thinking about mirror images. We then watched a short video about chirality (below):
Students then received the Lesson 47 Worksheet, working in pairs to model the compounds using the class set of molecular modeling kits. The worksheet concluded with students hypothesizing whether L-carvone will smell like D-carvone, and then testing their hypothesis. For homework, students were assigned textbook questions 5-8.
Update: January 18
Given the challenging nature of the subject matter in Lesson 47, we used most of the class period to review the homework, build molecules, and discuss the relationship between isomers and chirality. Notes from the overhead are shown below:
Want more? Check out the blog post Perhaps looking-glass milk isn’t good to drink for an overview of Lewis Carroll, looking-glass milk, and L- and D-carvone. Want more? Joanna Shawn Brigid O’Leary from Rice University published an even more extensive investigation of how Lewis Carroll (author of Alice in Wonderland and Through the Looking Glass) weaved biochemistry into his fiction. Her paper (available as a PDF), WHERE ‘THINGS GO THE OTHER WAY’: THE STEREOCHEMISTRY OF LEWIS CARROLL’S LOOKING-GLASS WORLD is well worth the read. Perhaps it will even inspire students to read the book before the movie is released in theaters on May 27!
Phase, Size, Polarity, and Smell
Chapter 8 concluded with the Lesson 46 PowerPoint and Lesson 46 Worksheet. Lesson 46 brought together the various concepts needed to understand how molecules with certain properties can be detected by our noses, with our brain recognizing those molecules as having a specific smell. The ChemCatalyst asks students to model why perfume molecules can be smelled from across a room, but paper cannot (both placed near a sunny window). Examples of the explanation (students and teacher versions) are provided below:


After the lesson, students received the Chapter 8 Quiz Study Guide in preparation for the review tomorrow and the Chapter 7/8 quiz scheduled for next Tuesday.
Polar Molecules and Smell
We continued our study of polarity, this time exploring how the polarity of molecules might impact our ability to smell the molecule. Through the Lesson 45 PowerPoint, students learned that polar molecules are more likely to be detected by the nose as something with a scent although there are still polar molecules (like water) that do not smell. Students worked in pairs to cut out the molecules in the molecules handout and used the molecules to complete the Lesson 45 Worksheet. Students who would like to explore the polarity of molecules further are encouraged to visit the University of Colorado’s PhET molecule polarity simulation (or just click below).
Electronegativity Scale
After learning about the concepts of electronegativity and polarity in yesterday’s lesson, students learned how scientist Linus Pauling assigned electronegativity values to individual atoms as a measure of how strongly an atom attracts electrons.
The Lesson 44 PowerPoint includes a copy of the periodic table with electronegativity values for each element. It also explains the difference in electronegativity between covalent bonds (0.5 and less), polar covalent bonds (between 0.5-2.1), and ionic bonds (greater than 2.1). The Lesson 44 Worksheet provides students with the opportunity to calculate the electronegativity difference between two atoms in a molecule and to use that information to determine the type of bond that is present between the two atoms. For homework, students are assigned questions 1-5 (odds).
Students were curious about new elements in the periodic table, so below is an article for the curious to learn more about the predicted “Island of Stability” and the theoretical upper limit in atomic number for the periodic table:





You must be logged in to post a comment.