In the first lesson of Chapter 14, students were introduced to the concept of LD50 (the dose of a compound that is lethal to 50% of the population). As our entry task, students considered the following:
Which substance do you think is most toxic to you –
- Alcohol (ethanol, C2H6O)
- Aspirin (salicylic acid, C7H8O3)
- Arsenic (III) oxide (As2O3)
Explain your thinking
We then identified the LD50 values for all three substances using the Lethal Doses Handout. We discussed the meaning of LD50 (the lethal dose at which 50% of rats die after exposure to a given amount of a substance, often expressed in mg/kg) and students were shown a Material Safety Data Sheet (MSDS) for water (toxic at >90 mL/kg), providing students with evidence that all substances are toxic at high enough levels. Students then received the Lesson 74 Worksheet to work on during class time.
Notes from class:

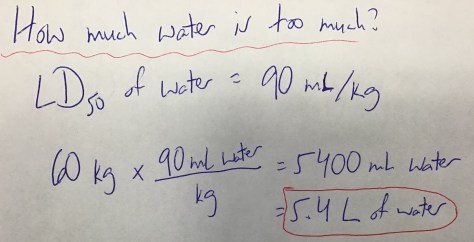
Extend Your Learning!
Wondering about how scientists define the kilogram? Turns out the way we define the kilogram just changed late last year. Read about it at PBS.org or watch the video below:
Homework:
- Read Lesson 74 in the textbook. Login via hs.saplinglearning.com and enter your username and password:
- Username: wahps****s-####### (**** = first 4 letters of your last name and ####### = student number). Remember to include the dash between s and #.
- Password: S-####### (the S must be capitalized)
- Work through the practice problems at the end of Lesson 74.
- Please ask questions about anything from Lesson 74 you do not yet fully understand.
Opportunities For Help Outside of Class:
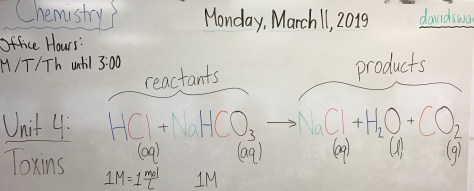
- Mr. Swart’s office hours: Mondays, Tuesdays, and Thursdays after school until at least 3:00 (later with advanced notice)
- Use the vast number of resources available on this website – check each lesson!
- Email Mr. Swart for help if staying after school is not possible
- Form a study group with other students to review concepts from class
- Attend MASH after school (M/T/Th in Library)
- Attend Math Lab after school (T/Th in Room 124)


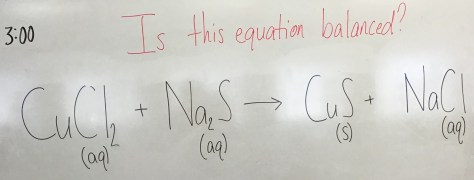
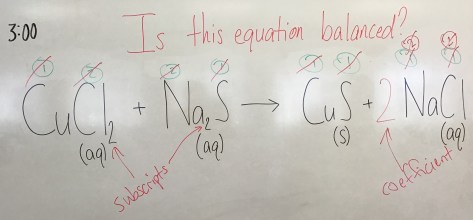
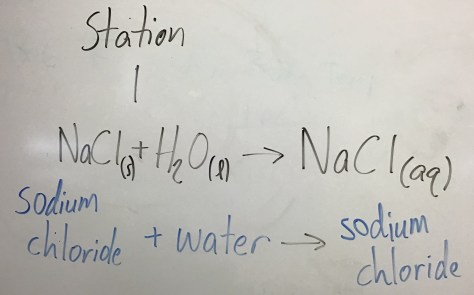

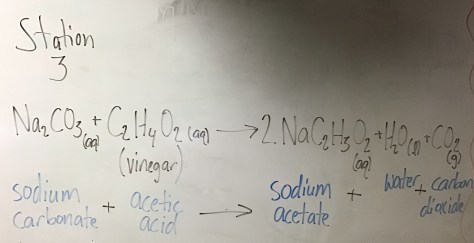





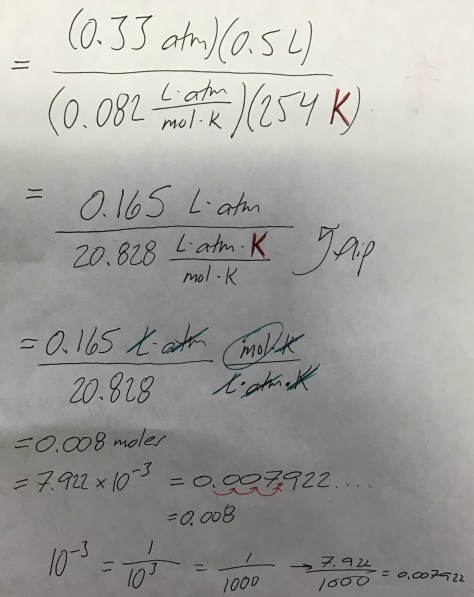

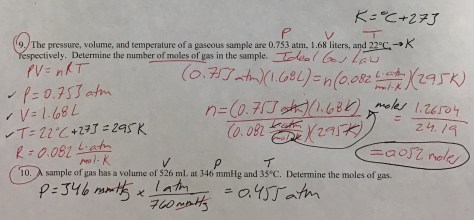
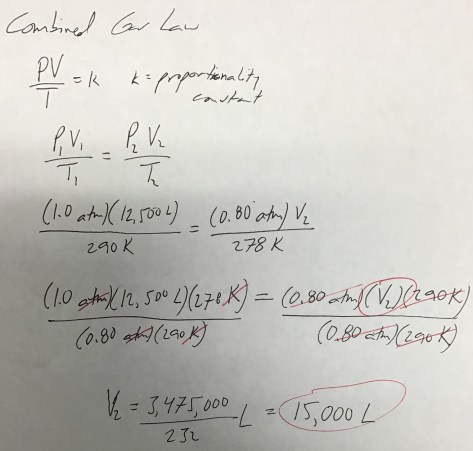





You must be logged in to post a comment.