After concluding the chapter 13-15 test last week, we used our class period today to work through the logistics of the next few weeks (an accounting of how many students are taking AP tests and/or are attending field trips) and then focused on student feedback about how to invest our class time. One theme that came up repeatedly was the application of chemistry to the environment through project work. To honor that feedback, students were tasked with reading and analyzing an article released this morning by CNN titled “Is Nature Over? Maybe.” After a class discussion (class notes below), we concluded the class period with the TED Talk below by George Monbiot on the concept of rewilding. The intent is to push students beyond familiar and comfortable solutions to the question: What can we do climate change?
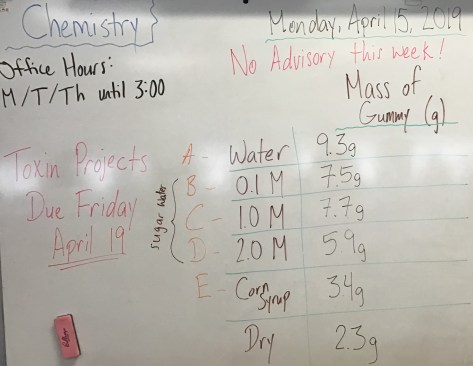
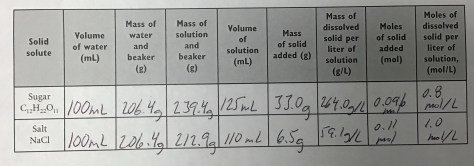
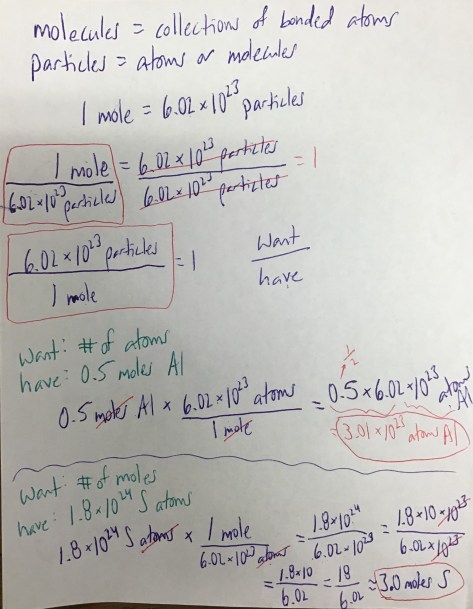
Class Notes:







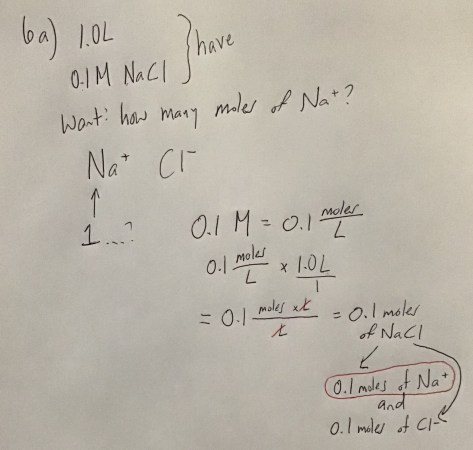
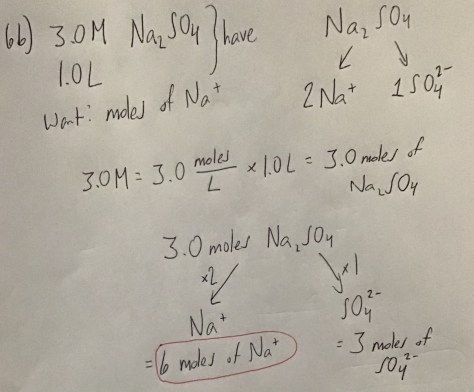

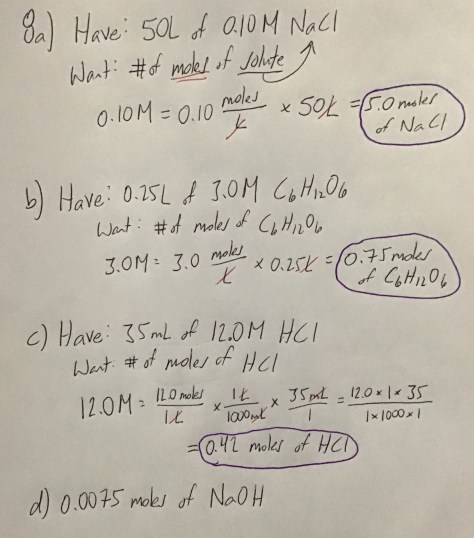
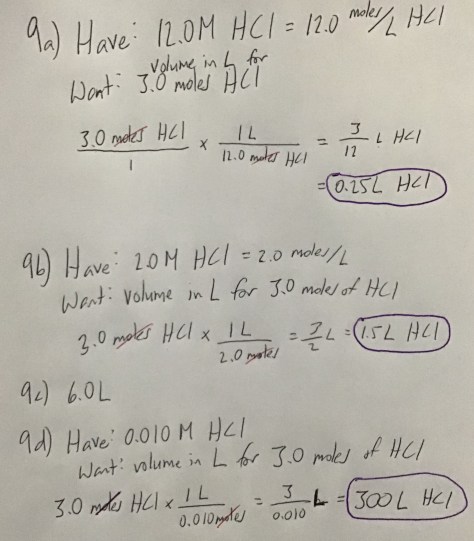










You must be logged in to post a comment.