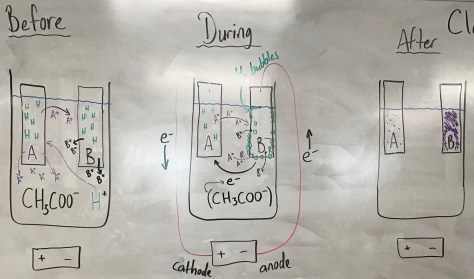
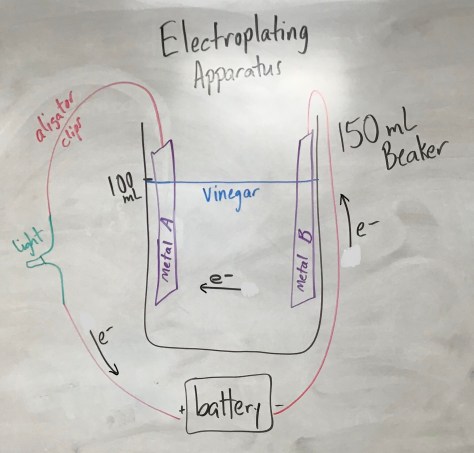
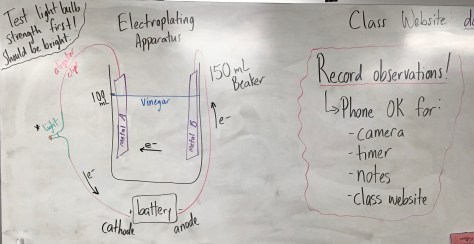
We continued our work from yesterday, beginning with the Lesson 29 PowerPoint. After preparing for the lesson, students worked through the Lesson 29 worksheet which included another “wafting” activity in which three additional scents were provided for students to smell and connect with molecular formulas. Students learned that two molecules can have the same molecular formula but smell very differently. One compound smelled like rum extract, while an isomer of that compound smells like stinky cheese. The compounds are isomers because they have the same chemical formula but different structural formula. The activity further enabled students to make connections between compound names, molecular formulas, structural formulas, and smell.
List of samples and songs:
- Tube F: apricot extract (sweet) – I Love Rock ‘n’ Roll
- Tube G: butyric acid (putrid) – Don’t Worry Be Happy
- Tube H: rum flavor extract (sweet) – Jack & Diane
Homework:
- Read Lesson 29 in the textbook. Login via hs.saplinglearning.com and enter your username and password.
- Write notes for Lesson 29 and work through the practice problems at the end of Lesson 29.
- Please ask questions about anything from Lesson 29 you do not yet fully understand.























You must be logged in to post a comment.